The Patient Connection July 2017
July 20th, 2017
•
47

Launch your research study apps within minutes.
Categories:
Tags:
#clinicalresearch ,
#digitalhealth ,
#MobiClin2018 ,
#scopesummit ,
15 years ,
2016 ,
ACRP2017 ,
active task ,
active task on ios ,
active tasks on android ,
Adventure ,
agentic ai ,
agentic ai in clinical trials ,
agentic ai in patient recruitment ,
AI ,
AI in Clinical Trials ,
AI in patient recruitment ,
AI-driven Patient recruitment tools ,
AICPA SOC 2 ,
ALS ,
Alzheimer's disease ,
American Heart Month ,
amia ,
AMIA 2016 ,
amia 2017 ,
AMIA2016 ,
AMIA2017 ,
amputee pain ,
Android ,
android apps ,
android research study apps ,
Android ResearchKit ,
app builder ,
app surveys ,
appbakery ,
apple active task ,
Apple ResearchKit ,
apps ,
artemis II ,
Artificial Intelligence ,
artificial intelligence in clinical trials ,
ASCO 2017 ,
ask Dory ,
astronaut data ,
astronaut displays ,
astronaut health ,
astronaut interaction ,
astronaut safety ,
autoimmune awareness month ,
Ax-4 ,
Ax-4 Mission ,
axiom mission 4 ,
Axiom Space ,
barriers in mhealth ,
BCI in space ,
BigData ,
biomedical data collection ,
bleeding disorder ,
bleeding disorder awareness ,
blood cancer awareness ,
brain health ,
brain initiative ,
brain-computer interfaces ,
breast cancer clinical trials ,
Cancer ,
cancer immunotherapy ,
Cancer in LEO-3 ,
cancer moonshot ,
cancer novartis ,
Cancer treatment ,
car t cancer ,
car t clinical trials ,
car t durgs ,
car t pharmas ,
CAR-T cell therapy ,
car-t immunotherapy ,
cardiovascular health ,
carekit ,
Carl June ,
Celiac disease ,
celiac disease awareness ,
cellectar ,
centerwatch ,
cervical cancer awareness ,
cervical cancer clinical trials ,
cervical health ,
challenges mhealth adoption ,
chatbots ,
clinical ,
clinical research ,
clinical trial ,
clinical trial awareness ,
clinical trial digital tools ,
clinical trial failure ,
clinical trial finder ,
clinical trial losses ,
clinical trial management ,
clinical trial management system ,
clinical trial management systems ,
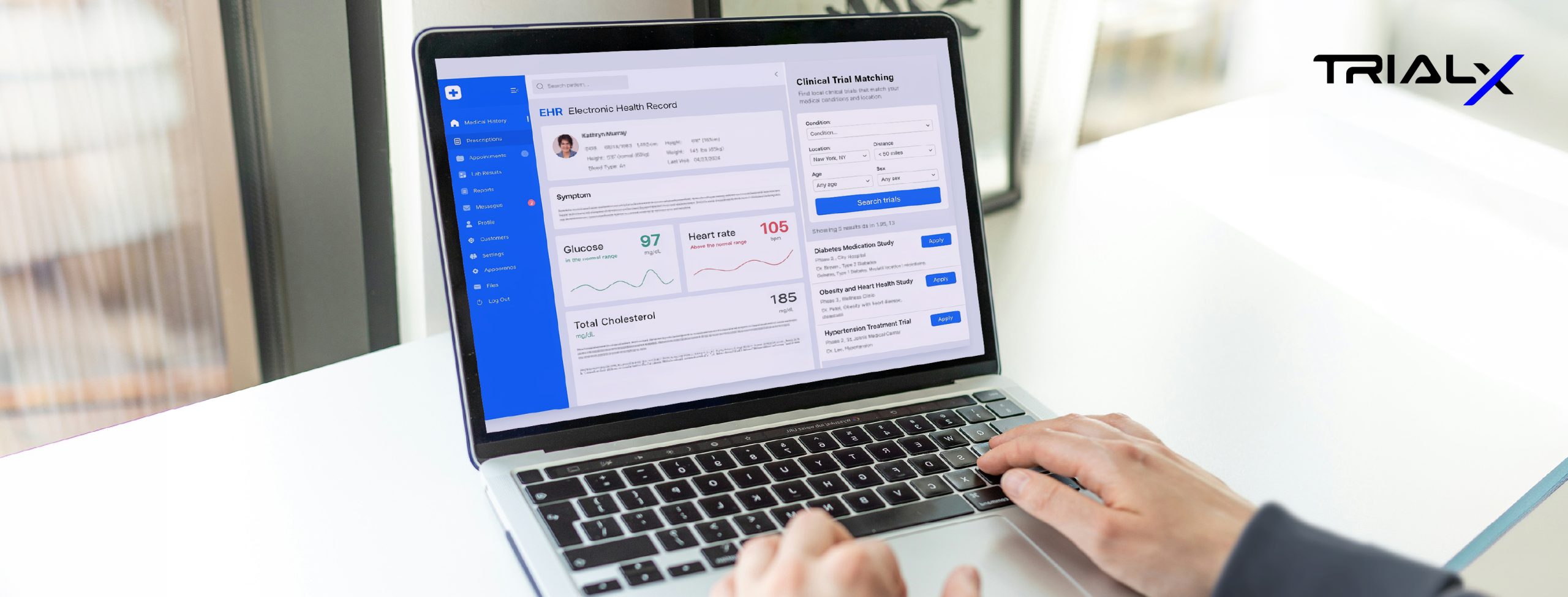
Clinical Trial Matching ,
clinical trial participation ,
clinical trial software ,
clinical trial sponsors ,
clinical trial website builder ,
clinical trials ,
clinical trials eligibility criteria ,
Clinical Trials in Space ,
clinical trials participation ,
clinical trials recruitment ,
clinical trials upenn ,
clinicalresearch ,
clinicaltrials ,
clinicaltrialvolunteers ,
commercial astronauts ,
Company Retreat ,
conference 2016 ,
conference 2017 ,
conference 2023 ,
consent under gdpr ,
Coronavirus ,
covid talk series with Penn Medicine ,
covid trials ,
COVID-19 ,
COVID-19 Cure ,
COVID19 ,
covid19 clinical trials ,
COVID19 therapy ,
COVID19 vaccine ,
Creative writing ,
crew health monitoring ,
crossplatform apps ,
ctms ,
ctms trialx ,
Cure ,
curetalks@penn ,
data accuracy ,
data privacy ,
data quality ,
DCT ,
Decentralised clinical trials ,
decentralized clinical trial ,
decentralized research ,
decentralized trials ,
DEI Toolkit ,
depression ,
depression clinical trials ,
depression research ,
DIA ,
DIA 2016 ,
DIA2017 ,
diabetes clinical trials ,
diet and lifestyle ,
digital patient engagement ,
disruptive innovations ,
diversity ,
diversity in clinical trials ,
DIY appmaking platform ,
DIY study apps ,
dpharm ,
DPharm 2016 ,
Dpharm2016 ,
dpharm2017 ,
drug development ,
drugs in 2016 ,
e-consent ,
eCOA ,
EHR ,
electronic Clinical Outcome Assessments ,
electronic displays in space ,
electronic health records (EHRs) ,
electronic patient reported outcomes ,
ePRO ,
equity ,
equity in clinical trials ,
EVA and Spacesuits ,
EXPAND database ,
extreme environment remote data collection ,
facebook ,
fda ,
FDA approvals ,
fda drug approval process ,
fda drug approved in 2016 ,
FDA perspective ,
Federal data ,
finger tapping researchkit ,
fNIRS ,
games ,
gamification ,
GDPR ,
genome hacking ,
global clinical trials ,
gregory dumanian ,
Hackathon ,
Halloween ,
healthcare ,
healthy controls ,
healthy subjects ,
healthy volunteers ,
HERMES platform ,
HIPAA compliance ,
human atlas ,
human cell atlas ,
human factors in space ,
iconnect ,
iconnect clinical trial ,
iConnect features ,
IDY ,
Immunotherapy ,
immunotherapy cancer ,
inclusion ,
inclusion in clinical trials ,
inclusivity ,
indiana clinical trials ,
infectious disease ,
informatics ,
informed consent ,
Inspiration ,
international space station ,
internationalization in research study apps ,
investment ,
iOS ,
ios apps ,
ISRO Space Research ,
key fda drugs ,
language barrier ,
limb loss pain ,
lung cancer awareness ,
Lyme ,
lyme diagnosis ,
Lyme disease ,
lyme disease management ,
Lyme disease tracker app ,
lyme symptoms ,
march is national nutrition month ,
medical apps ,
medical breakthrough ,
medical imaging ,
Mega projects ,
mental health ,
mental health clinical trials ,
mhealth ,
mhealth adoption ,
mhealth in clinical trials ,
mhealth precision medicine ,
mhealth tools ,
microgravity ,
Microgravity Effects on Muscles ,
microgravity research ,
milestone ,
minority participation ,
Mitochondrial Function in Space ,
mobile apps ,
mobile clinical innovations ,
mobile health technology in engagment ,
mobile in clinical trials ,
Mobile Research ,
Mobile Research Apps ,
mobile research study app ,
mobile research study apps ,
mobileclin2017 ,
mobileclin2022 ,
mrna ,
mrna vaccine ,
multi-language apps ,
multi-language research apps ,
multilingual support ,
Muscle Loss in Space ,
my clinical trial story ,
Myogenesis Experiment ,
NASA spacesuit audit ,
national nutrition month ,
neurogenic pain ,
new data privacy policy ,
new release ,
new video format ,
nobel prize ,
nobel prize laureates ,
northwestern university ,
novel coronavirus ,
nutrition awareness ,
nutrition research ,
NYU ,
nyu clinical trials ,
online talk shows ,
orphan drugs ,
ovarian cancer awareness ,
pain app ,
pancreatic cancer clinical trials ,
pancreatic cancer research studies ,
parkinsons ,
parkinsons awareness ,
patient ,
patient centric ,
patient centricity ,
patient engagement ,
patient engagement in clinical trials ,
patient generated health data ,
patient involvement ,
patient involvement in clinical trials ,
patient participation ,
patient portals ,
Patient Recruitment ,
patient referrals ,
patient reported outcomes ,
patient safety ,
patient-reported outcomes ,
patientengagement ,
patientfocussed ,
patients ,
patienty privacy ,
penn clinical trials ,
Penn Medicine ,
personal health devices ,
personal health information gdpr ,
Philadelphia ,
physiology and medicine ,
precision medicine ,
privacy and security ,
prompts ,
rare disease ,
rare disease awareness ,
rare disease day ,
real-time data ,
Recruitment Tools ,
regulatory compliance ,
remote data ,
remote data collection ,
remote diagnostics ,
Remote Health Data Collection ,
Remote Research Data Collection ,
remote research data collection platform ,
research ,
research apps ,
research study apps ,
Research Volunteers ,
ResearchDroid ,
researchkit ,
researchkit active task ,
researchkit active task list ,
researchkit active tasks ,
researchkit android ,
researchkit apps ,
rett syndrome trials ,
Sars-Cov-2 ,
SARS-COV2 ,
SCOPE2018 ,
scope2023 ,
SCOPE2025 ,
scopesummit ,
security compliance ,
sellas life sciences ,
sickle cell disease ,
sleep in space ,
sleep self management ,
sleep study ,
sleep tracking ,
sleep tracking app ,
small biotech companies ,
SOC 2 ,
SOC 2 Compliance ,
Social Media ,
space clinical research ,
Space Health ,
space health research ,
space health technology ,
space medicine ,
space neuroscience ,
space oncology ,
space research ,
spaceflights ,
spacehealth ,
spaceresearch ,
Stem Cell Regeneration ,
study apps ,
Team building ,
Team Outing ,
Team Retreat ,
Telemetric Health AI ,
The Opportunity Project ,
thyroid awareness month ,
tokenization ,
TOP Health Challenge ,
tophealth ,
tophealthchallenge ,
track sleep ,
trial participation and engagment ,
trialspace ,
trialx ,
trialx iconnect ,
TrialX Space Health Systems ,
triple-negative breast cancer ,
trump fda ,
tsc ,
tuberous sclerosis complex ,
twitter ,
U.S. Elections ,
ultrasound ,
University of Pennsylvania ,
unsung but impactful ,
Upenn ,
vaccine trials ,
Virology ,
voices of clinical research ,
volunteer registry ,
wearable devices ,
wearables ,
Webinars ,
Website Builder ,
weight loss clinical trials ,
weight management clinical trials ,
White House ,
whitepaper ,
world cancer day ,
World Health Day ,
world parkinsons day ,
world yoga day ,
year end review ,
year2022 ,
0
19418