TrialX at SCOPE 2026: Simplifying Patient Recruitment, Spotlighting Clinical Research Heroes — Coffee, Conversations & Key Insights

Team TrialX at SCOPE 2026: Sharib Khan (Co-founder & CEO), Wes Martz (Senior Director of Client Services), Beth Dickson (VP, Business Development), and Brandon Ramos (Sales Development Representative)
SCOPE 2026 once again brought together leaders across clinical operations, digital health, patient engagement, and data innovation to shape the future of clinical research. This year’s conversations reflected a clear shift. The industry is no longer asking if transformation is needed. The question now is how fast it can be implemented responsibly and effectively. From AI-driven workflows to more strategic patient recruitment, these were the key themes that emerged—and how TrialX is contributing to this evolving, more patient-centric clinical research landscape.
Key Highlights
1. AI Becomes Core to Clinical Trial Operations
At SCOPE 2026, AI was no longer positioned as a future concept. It was operational reality. Sponsors and CROs are embedding AI into patient matching, protocol feasibility, enrollment forecasting, data monitoring and site optimization. The conversation has shifted from isolated pilots to scalable, compliant systems with measurable ROI. Sessions on Agentic AI and keynote discussions on AI adoption reinforced a common theme. AI must be governed, auditable, and aligned with real-world workflows. At TrialX, we see this evolution firsthand. Our AI-powered trial matching is built for precision, transparency, and human-centered engagement.
2. Patient Recruitment as a Strategic Priority
Recruitment is being redefined from a tactical challenge to a strategic function embedded early in trial design. Leaders emphasized multi-channel outreach, community-based engagement, diversity and equity strategies and real-time analytics for predictability. The tone across sessions was clear. Recruitment cannot be an afterthought. It must be integrated into feasibility, protocol design, and site strategy from the start. Sponsors are moving toward centralized, end-to-end recruitment ecosystems, and TrialX supports this shift through:
- High quality trial matching
- Volunteer registries
- Integrated patient engagement platforms
3. Hybrid Models Are the New Standard
The industry has moved beyond debating decentralization. Hybrid trials are now the default. Combining site oversight with digital flexibility to reduce patient burden while protecting data integrity. Remote data capture, eConsent, digital endpoints, and connected devices are no longer optional enhancements. They are foundational. SCOPE sessions on digital measures, eSource, and integrated endpoints reinforced that the next challenge is workflow compatibility. TrialX’s work in remote clinical research infrastructure, including space health initiatives, reflects this broader move toward borderless, resilient clinical research.
4. Integration Is the Next Competitive Advantage
As trial ecosystems grow more complex, interoperability has become essential. Sponsors are demanding seamless data flow, unified oversight dashboards, reduced operational redundancies and fewer fragmented vendor stacks. The message from SCOPE was consistent across panels and networking discussions: technology must simplify workflows, not fragment them further. Smarter integration, not more tools, will define the next phase of clinical innovation.
TrialX Leadership Insights from SCOPE 2026
As proud sponsors, the TrialX team was at Booth #225— Sharib Khan (Cofounder & CEO), Wes Martz (Senior Director of Client Services), Beth Dickson (VP, Business Development), and Brandon Ramos (Sales Development Representative)—connecting with industry leaders and sharing how our enterprise patient recruitment platform and remote data solutions support modern trials.

For us at TrialX, SCOPE was not just about booth traffic, but about reinforcing a critical reality: clinical trial success in 2026 depends on reliable, integrated patient engagement and enrollment infrastructure — not disconnected tools. The enrollment landscape is increasingly complex. Sites are stretched, sponsors face accelerating timelines, and regulators continue to prioritize transparency and participant-centricity. Across discussions, a strong consensus emerged: enrollment can no longer be treated as a standalone campaign, but as a core, measurable part of the broader clinical research ecosystem.
At our booth, conversations focused on:
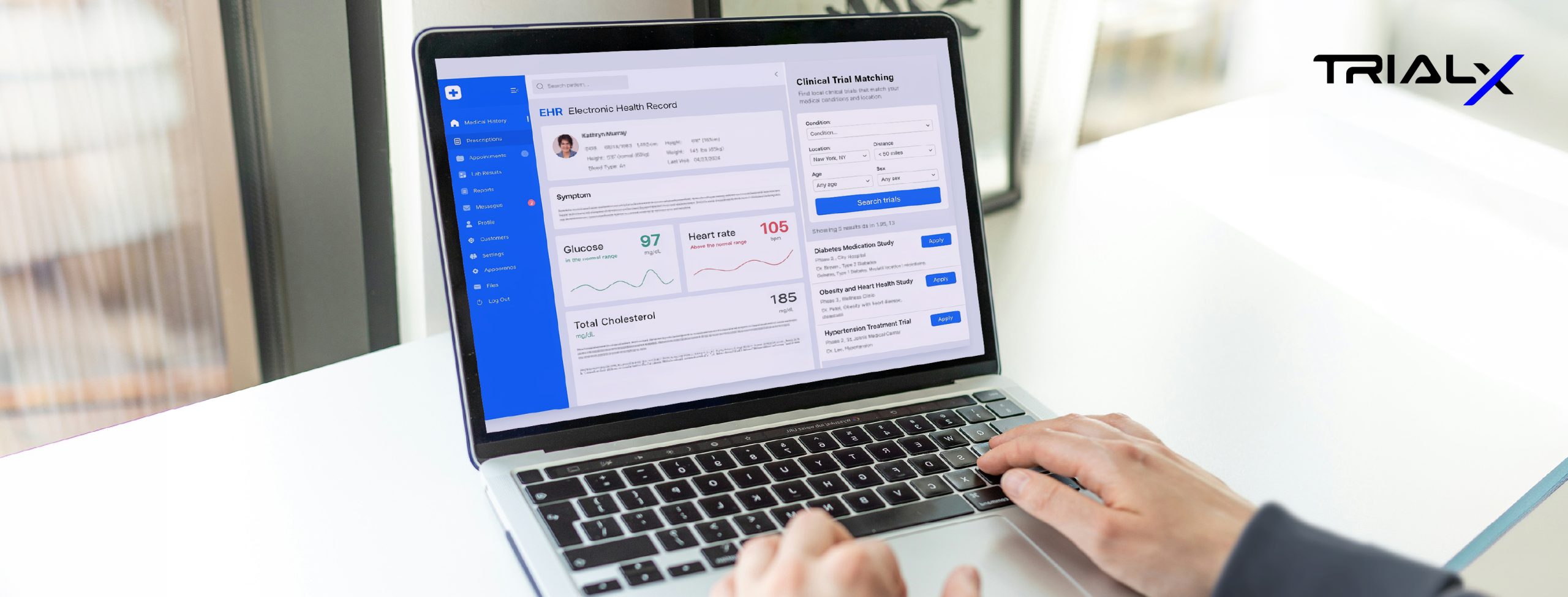
- Operationally Reliable Trial Finders: Many organizations are still relying on static directories or internally built tools. What we discussed repeatedly is the challenge of maintaining accuracy, searchability, compliance, and real-time performance at scale. TrialX’s platform powers dynamic trial discovery and referral workflows that connect interest to actual enrollment outcomes — not just clicks.
- Enrollment as Infrastructure, Not a Tactic: Recruitment is often treated as a short-term service layered onto a study. Increasingly, leaders are recognizing the need for persistent registries, reusable prescreeners, analytics dashboards, and site-connected workflows that compound value over time.
- Analytics That Matter: A recurring question at SCOPE: “Can you show us what’s actually converting?” Visibility into referral flow, site follow-up, patient drop-off points, and campaign performance is no longer optional. Data transparency is becoming foundational to how sponsors and CROs evaluate partners.
- Participant-Centered Design: As decentralized and hybrid trials continue to evolve, reducing friction for participants is critical. From intuitive prescreeners to compliant communication pathways, engagement must feel simple on the front end — even if the infrastructure behind it is sophisticated.
As regulatory expectations mature and competition for participants intensifies across the globe, organizations are realizing that enrollment performance is directly tied to the strength of their underlying technology and operational processes. Sponsors were not looking for more point solutions. They were looking for systems that reduce friction across recruitment, engagement, and data workflows.

Across meetings with sponsors, CRO leaders, patient engagement executives, and technology partners, one message was consistent: Recruitment must be continuous. Remote data must be realistic. AI must be trusted. Workflows must reflect how trials actually run. These were not isolated talking points. They were reinforced across keynotes, awards sessions, and cross-functional panels.
From Discussion to Execution
SCOPE Summit 2026 highlighted a clear industry shift from digital experimentation to operational execution.
Across Day 1 sessions, including the INSPIRE Agentic AI Summit by Maxis AI and WCG’s industry roundtable, leaders emphasized embedding AI and integrated data strategies directly into clinical operations to improve trial quality, feasibility, and predictability. Workshops on AI digital maturity and modernizing informed consent reinforced that scaling AI requires governance, ethical safeguards, and leadership readiness.
Operational efficiency was another major focus. Deloitte and PA Consulting addressed persistent inefficiencies in trial finance, pipeline delivery, and fragmented workflows, calling for unified data structures and adaptable operating models. Meanwhile, a powerful fireside chat on patient experience reminded attendees that empathy, transparency, and real-world practicality must remain central to trial design.
Day 2 and 3 keynotes expanded the conversation to acceleration and alignment. Leaders from Merck, Medidata, AstraZeneca, AWS, Novartis, Pfizer, Amgen, and AbbVie discussed therapeutic specialization, data minimization to reduce patient burden, AI-enabled predictive analytics, and privacy-preserving real-time data access. Panels explored whether radical acceleration is possible. The consensus: compression depends on smarter upfront planning, cross-industry collaboration, and AI that enhances productivity without compromising clinician–patient trust.
The final day reinforced that clinical development cannot operate in silos. A recurring theme throughout the summit was clear. Technology readiness is no longer the constraint. Organizational change, integration, and human-centered implementation will define the next era of clinical research.
Fueling Conversations at SCOPE
Sometimes the most practical insights happen outside the formal agenda. Our Coffee & Dessert Break became a natural gathering point. Between sessions, attendees connected over conversations on feasibility, hybrid trial execution, AI governance, and enrollment predictability.
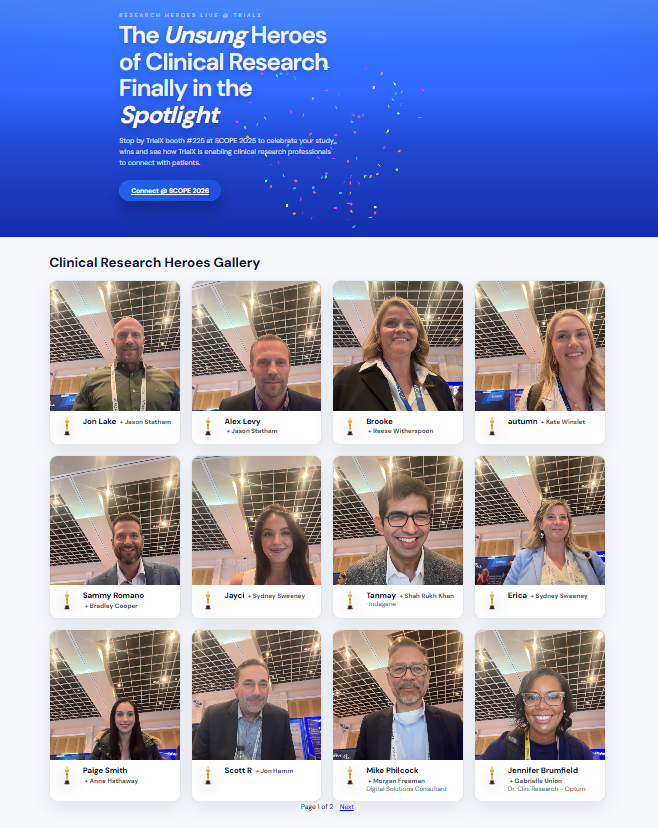
Spotlighting Clinical Research Heroes
And yes, our celebrity face mashup activity drew a crowd, adding a fun spotlight moment to meaningful conversations.
Clinical research is possible largely because of the people behind the scenes. As a small gesture to honor the contributions made by them, at SCOPE 2026, we launched the Clinical Research Heroes gallery at Booth 225. Attendees selected their “celebrity alter ego,” and our AI generated personalized spotlight portraits in a few seconds. The result was more than a mashup. It was a recognition moment.
Checkout our Clinical Research Heroes Gallery now live here: 👉 https://clinicalresearchheroes.trialx.com/
The experience reflected a broader truth echoed throughout SCOPE – Behind every protocol, endpoint, and dataset is a person driving progress.
The Takeaway
SCOPE 2026 confirmed that the industry is no longer experimenting with transformation. It is operationalizing it. The organizations that will move fastest are those that:
- Embed recruitment early
- Integrate remote data capture intelligently
- Treat AI as infrastructure
- Reduce site burden
- Design workflows around operational reality
At TrialX, these are not future ambitions. They are systems actively being built and deployed.
For those we connected with in Orlando — thank you for the thoughtful conversations. For those who couldn’t attend, we’d welcome the opportunity to share how TrialX supports sponsors, CROs, advocacy groups, and sites with scalable enrollment solutions that drive measurable outcomes. The future of clinical research won’t be built on fragmented systems. It will be built on connected, intelligent platforms that turn patient interest into participation — reliably.
Looking forward to continuing the conversation at the upcoming Patients as Partners 2026 conference. Join our panel discussion on – Innovative solutions and technologies that enhance patient-enabled trials and services – on March 24th at 3:50 PM.