Improving Global Patient Recruitment and Clinical Trial Access – TrialX Leadership Perspectives & Key Highlights of Patients as Partners 2026
Key Highlights Patients as Partners 2026
1. AI Moving from Concept to Clinical Reality
Discussions showcased real-world AI applications—from patient digital twins to AI-assisted protocol development. The emphasis was on responsible implementation, governance, and measurable impact to enhance trial design, access, and patient experience.
2. Operational Realities Took Center Stage
Industry leaders openly addressed challenges such as budget constraints, organizational restructuring, and regulatory complexities. Practical insights focused on sustaining patient engagement while navigating legal and compliance requirements.
3. Measuring Impact Is Now Essential
Organizations presented data-driven outcomes, including faster site activation, fewer protocol amendments, improved retention, and enhanced diversity. Demonstrating ROI and tangible value from patient-centric initiatives emerged as a shared priority.
4. Inclusion Is Becoming Structured and Accountable
Diversity and representation efforts are evolving into measurable, regulatory-driven strategies. With growing emphasis from global regulators, inclusion is no longer aspirational—it is expected, tracked, and operationalized across clinical research.
PASP 2026 – TrialX Leadership Perspectives

The biggest takeaway at the Patients as Partners in Clinical Research conference: patients aren’t just participants — they’re essential partners in shaping better clinical trials and future treatments.
However, one theme echoed across sessions: making trials more accessible, inclusive, and easier to navigate isn’t optional anymore, it’s expected. While the industry has talked about patient centricity for years, the shift now is undeniable. Patients are demanding better access, simpler participation, greater transparency, and a true voice in their care. Notably, many patient advocates highlighted that trial endpoints, while clinically meaningful, don’t always reflect the outcomes that matter most to patients, such as symptom relief or quality of life.
Emerging technologies like AI will help address some of these challenges, but we can’t lose sight of what matters most. Clinical research is, first and foremost, human. The stories shared throughout the conference were a powerful reminder: disease doesn’t discriminate — and neither should we.
Our Senior Director of Client Services, Wes Martz, joined Krista Goedel from Sanofi on a panel to share innovative approaches to improving global patient recruitment and access to clinical trials. And our Co-founder & CEO, Sharib Khan, brought good energy (and balance) to the event with a fun, interactive yoga session, adding a refreshing dimension to the clinical research experience.
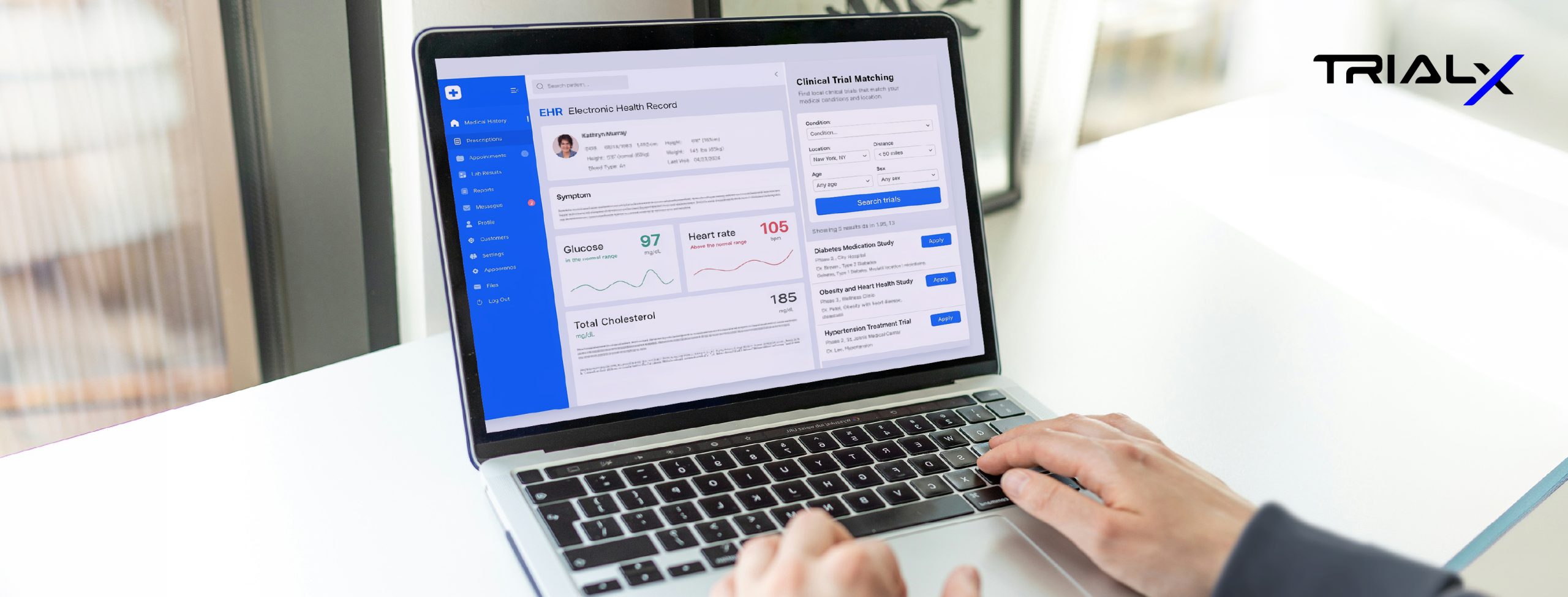
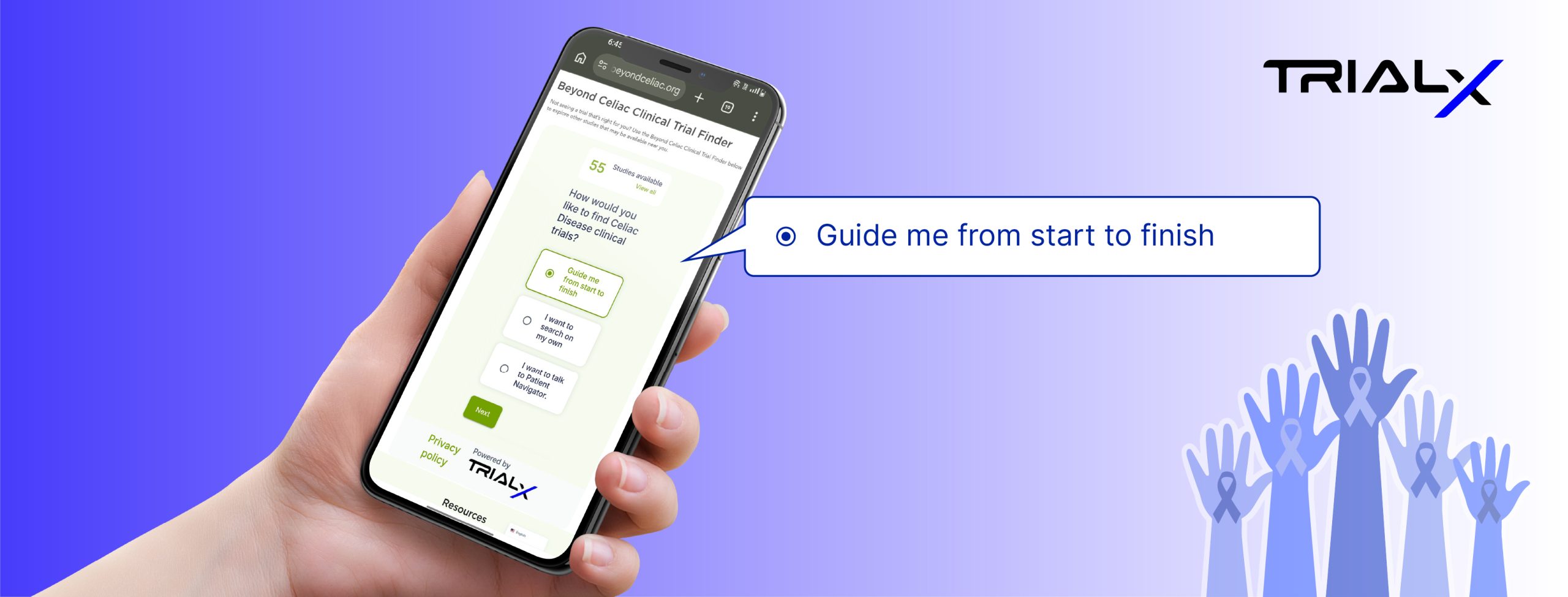
Building Scalable, Patient-Centric Clinical Trial Websites to Improve Global Recruitment and Access.
Our Innovative Solutions session with Wes and Krista, focused on a critical, and often overlooked, part of patient engagement: the digital front door. For many patients, the first interaction with a clinical trial begins online. Yet too often, that experience is shaped more by compliance requirements than by usability—resulting in static pages, complex navigation, and unclear next steps. In many cases, patients disengage before recruitment teams even know they were there.

Wes and Krista shared an alternative approach—designing purpose-built, patient-facing trial websites around how people actually search, read, and make decisions. Drawing from experience scaling global trial platforms across 18+ countries, they highlighted what it takes to balance localization, accessibility, regulatory requirements, and a consistent user experience without compromising usability.
This included building multilingual experiences, enabling regional flexibility while maintaining consistency, and creating clearer pathways from discovery to enrollment. Just as importantly, the session addressed what works, what doesn’t, and how patient-facing platforms need to evolve over time to better support global recruitment.
The session reinforced an important point: digital infrastructure plays a foundational role in shaping the patient experience. When designed well, it can significantly improve access and engagement. When overlooked, it can become an early barrier.
A Short Recharge: 5-Minute Yoga Session Hosted by TrialX

Our CEO and Co-founder, Sharib Khan, isn’t just a tech visionary; he’s also a passionate yoga enthusiast and certified yoga guru. In between sessions, attendees were invited to pause for a short, guided yoga break hosted by Sharib. It was a brief pause in a packed agenda, yet a meaningful opportunity to reset and recharge—one that attendees truly appreciated.



In summary, PASP 2026 highlighted an industry advancing toward responsible AI adoption, data-driven decision-making, operational resilience, and equitable, patient-centered innovation. It reinforced something we strongly believe at TrialX: the infrastructure behind patient engagement matters as much as the intention. Recruitment, access, digital experience, and data are not merely operational considerations—they determine whether patients can find and participate in clinical trials.
We left with new connections, fresh perspectives, and a clearer view of where the field is headed. We look forward to continuing the conversation in London at Patients as Partners Europe.
Interested in learning more about how TrialX supports patient-centric clinical trial recruitment and digital access? Get in touch.