How TrialX Patient Recruitment Management Solution Addresses Language Barriers in Clinical Trial Participation
Clinical trials are becoming increasingly global. Studies now run across countries, involve participants from different cultures, and collect data from multiple healthcare systems. In principle, this should support broader representation and stronger evidence. But in practice, one barrier continues to limit who can actually participate: language.
Language affects many parts of a clinical trial. It influences who learns about a study, who is considered eligible, how clearly consent is understood, and whether participants stay engaged through completion. Participants with limited English proficiency (LEP) are more likely to be excluded, misunderstand study requirements, or disengage early—often without intent from research teams. While 91.5% of researchers say it is important to include participants with LEP, 72.6% admit they exclude them at least some of the time. This gap between intention and practice continues to affect diversity, equity, and the reliability of trial outcomes.
Where Language Barriers Appear in Clinical Trials
Language barriers emerge at multiple points in a participant’s trial journey—across time, settings, and moments that are critical to enrollment and retention.
In an interview published by Science, physician-scientist Edward Garon recalled identifying an ideal candidate for a lung cancer trial early in his career. “I was very excited,” he said. “There was just one problem: The patient only spoke Spanish.”
To enroll, the patient had to “comprehend—and sign—dozens of pages of documents, agreeing that she understood the trial’s risks as well as its time and logistical commitments.” Securing translated consent materials required additional funding and coordination. Reflecting on the situation, Garon said, “I remember the anxiety I felt.”
Clinicians and public health professionals continue to report recurring communication challenges during:
- Initial screening and eligibility discussions, where misunderstandings can prevent otherwise eligible participants from enrolling
- Informed consent, which requires clear, culturally appropriate explanations of risks, procedures, and expectations
- Safety and adverse event reporting, where participants may struggle to describe symptoms accurately
- Ongoing engagement, when participants need timely clarification or support throughout the study
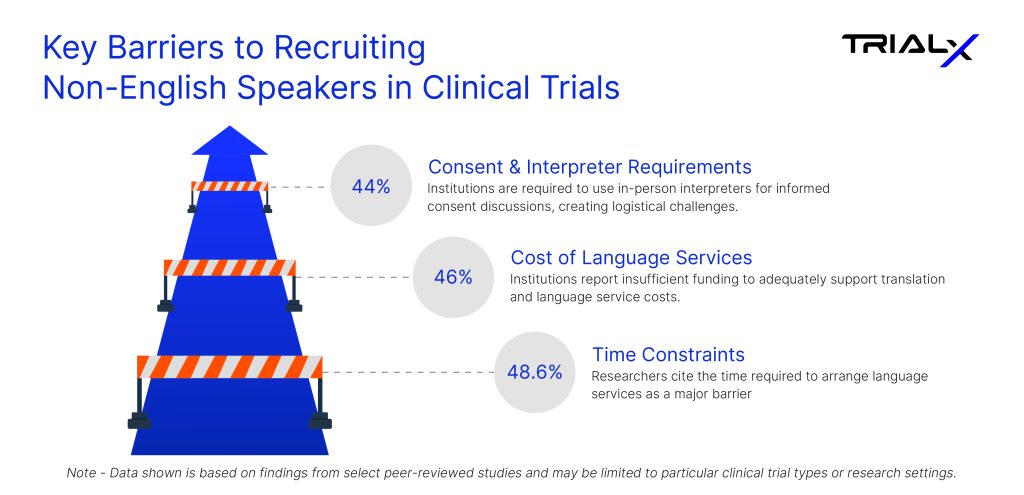
Why Language Barriers Persist in Clinical Trials
Time constraints: Nearly half of the researchers report that arranging interpretation or translation services requires additional time. In fast-moving trial environments, this can delay screening conversations, postpone consent discussions, or cause potential participants to lose engagement—particularly in decentralized or community-based studies where timing is critical.
Cost of language services: Professional interpretation and document translation add measurable expense to trial budgets. Smaller research sites and community clinics, which often serve linguistically diverse populations, may lack dedicated funding for these services, limiting their ability to consistently support multilingual participation.
Limited access to resources: Only a minority of research teams have access to in-house interpreters. Many depend on external vendors or scheduled appointments, which are not always available during real-time participant interactions. As a result, conversations may be shortened, rescheduled, or avoided altogether.
Informed consent challenges: Consent forms are typically lengthy, technical, and legally sensitive. Without reliable interpretation support, research teams may struggle to confirm that participants fully understand the study’s risks, procedures, and commitments. This can lead to delayed enrollment or, in some cases, exclusion to avoid compliance risk.
How TrialX Patient Recruitment Management Solution Is Addressing Operational Constraints In Global Trials
Research teams are widely aware of the need to include linguistically diverse populations, but operational realities often limit what is feasible. Time pressure, fragmented workflows, regulatory variation, and reliance on external language services make it difficult to support multilingual participation consistently across regions.
TrialX patient recruitment management solution is designed to help study teams by integrating multilingual and multicountry capabilities directly into recruitment and engagement workflows—supporting inclusion without adding unnecessary complexity.
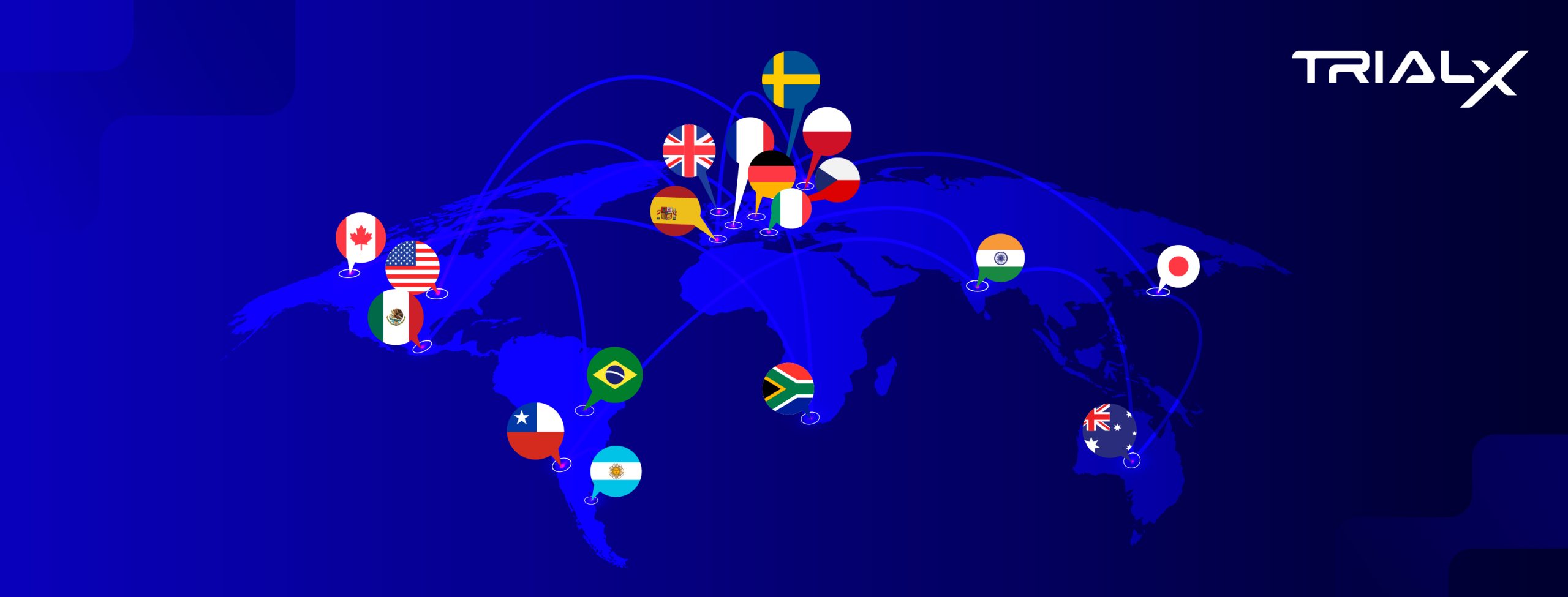
Recently, we supported one of the top-five pharmaceutical sponsors in a celiac disease clinical trial, where our platform enabled multilingual participant engagement and helped the study reach its enrollment goals. These capabilities currently support engagement in 14 languages across 18 countries, with expansion planned to 40 countries. We’re breaking down barriers to boost trial participation by:
- Providing Multilingual Support Across Countries
Language barriers often appear at the first point of contact—when participants encounter trial information that feels unfamiliar, unclear, or not intended for them. Clear communication across languages requires consistency throughout discovery, screening, and outreach.
TrialX supports multilingual engagement by enabling research teams to create localized study experiences and recruitment campaigns across 18 countries, with study experiences available in multiple languages, including:
- English — United States, United Kingdom, Australia, Canada, India, South Africa
- Spanish — Argentina, Chile, Mexico, Spain, United States
- Portuguese — Brazil
- French — France, Canada
- German — Germany
- Italian — Italy
- Hindi — India
- Japanese — Japan
- Czech — Czech Republic
- Polish — Poland
- Swedish — Sweden
- Afrikaans, isiXhosa, isiZulu — South Africa
Research teams can support multilingual participation through:
- Trial landing pages in local languages
- Multilingual prescreening and eligibility questionnaires
- Translated consent documents and study materials
- Recruitment messaging adapted by language and geography
Aligning study content and outreach with participant language preferences can help reduce early drop-off and support more consistent engagement across countries.
- Supporting Compliance in Multicountry Clinical Trial Operations
Running studies across countries involves differences in regulatory requirements and day-to-day operations. TrialX supports multicountry trial workflows through:
- Data privacy features aligned with frameworks such as HIPAA, GDPR, and region-specific requirements
- Time zone–aware scheduling and communication workflows
These capabilities help research teams operate across geographies while maintaining regulatory alignment and operational clarity.
Expanding Access Through Language
Language accessibility in clinical trials directly affects who is able to participate. When language barriers are reduced, trials become more representative, data become more reliable, and participants are better equipped to make informed decisions.
By enabling multilingual and multicountry capabilities, TrialX supports more inclusive trial design across global studies, helping research teams reach the right participants, in the right language, at the right time. Learn how TrialX can support your global study needs—Contact us here.