How TrialX AI-Powered Clinical Trial Matching Leverages EHR Data to Identify Eligible Patients Efficiently
Every day, healthcare systems generate enormous amounts of clinical data.
Diagnoses. Lab results. Imaging reports. Medications. Physician notes. Follow-up visits.
Most of this information is recorded in electronic health records (EHRs)—digital systems designed to support patient care and document clinical decisions over time. Clinical research, meanwhile, depends on detailed patient information to evaluate new therapies and improve treatment options. Yet identifying patients who may be eligible for clinical trials often requires research teams to manually review medical records and compare them against detailed eligibility criteria.
Across the industry, nearly 80% of clinical trials fail to meet their original enrollment timelines, and recruitment delays remain one of the most common reasons studies run over schedule and budget. Given the amount of clinical information already captured during routine care, researchers and health systems have increasingly explored whether existing EHR data could help support the process of identifying potential trial participants.
In practice, however, connecting everyday clinical documentation with structured clinical trial eligibility requirements is not always straightforward. Our AI-powered TrialX platform is designed to help address this challenge by leveraging EHR data to support more efficient patient–trial matching.
Why Clinical Records Don’t Easily Translate to Trial Eligibility
Electronic health records transformed healthcare by digitizing patient information. What once existed as paper charts became searchable medical histories that could be shared across departments and care teams.
This shift made it easier for clinicians to access patient data across visits, specialities, and health systems.
But EHRs were built primarily for clinical care—not clinical research.
A physician might document something like:
“Stage II breast cancer, currently on second-line therapy.”
For clinical care, that description is perfectly clear.
But clinical trial protocols often require far more detailed eligibility criteria, such as:
- Tumor size within a specific range
- No prior exposure to a particular therapy
- Specific biomarker thresholds
- Liver enzyme levels below a defined limit
- No history of autoimmune disease
All of this information may exist somewhere in the patient’s record, but it is rarely organized in a way that allows automatic trial matching.
The Reality of Identifying Eligible Candidates
Because of these structural differences, identifying eligible patients often falls to physicians and research coordinators manually reviewing medical records.
That process can be time-consuming in already demanding clinical environments.
As one physician respondent in a clinical trial referral survey by Drug and Device World described:
“It’s not just the search—making referrals is a headache. We’re often left with disjointed systems that don’t talk to each other.”
Data from multiple studies reflects this challenge across the industry.
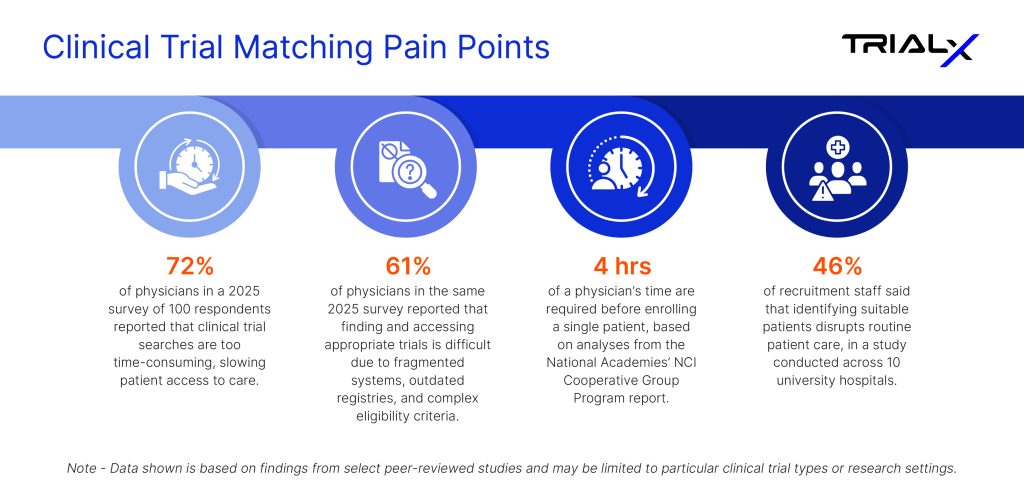
The problem, in other words, is rarely the absence of eligible patients.
It is the difficulty of identifying them efficiently.
What Research Shows
Researchers have explored how electronic health record data can support clinical trials more directly.
One well-known example is the Salford Lung Study, conducted across more than 80 general practices and 130 pharmacies in the United Kingdom. Instead of creating a separate research infrastructure, investigators used routine healthcare records to monitor outcomes for patients using a new inhaler for asthma and COPD.
Published in 2016, the study demonstrated that large-scale trials could operate within existing healthcare systems, with significant cost efficiencies. Highly automated EHR-supported trials were estimated to cost around $44 per patient, compared to roughly $2,000 per patient in more traditional trial designs.
In 2024, researchers from the National Institutes of Health’s National Library of Medicine introduced TrialGPT, an artificial intelligence system designed to help match patients with clinical trials more efficiently. The tool analyzes structured patient summaries—including age, medical conditions, and symptoms—and searches the ClinicalTrials.gov database to identify potentially relevant studies. The system then ranks clinical trials based on relevance and provides explanations showing how a patient’s medical information may align with each study’s eligibility criteria. In evaluation studies published in Nature Communications, TrialGPT retrieved approximately 90% of relevant trials and reduced the screening time by 42.6% in patient recruitment.
Another approach, the CrOss-Modal PseudO-SiamEse network (COMPOSE) framework, was developed by clinical informatics researchers studying automated patient–trial matching using EHR data. By modeling both patient medical records and clinical trial inclusion and exclusion criteria simultaneously, the framework improved the accuracy of automated trial matching in experimental testing.
These research shows how EHR data and AI are being used in real-world clinical trial matching, improving accuracy and streamlining recruitment.
How TrialX Connects Electronic Health Records to Eligibility Criteria Using AI-Powered Clinical Trial Matching
Our AI-powered TrialX platform supports more efficient patient–trial matching by leveraging EHR data. These capabilities help address several common recruitment challenges.
For research teams and study sponsors
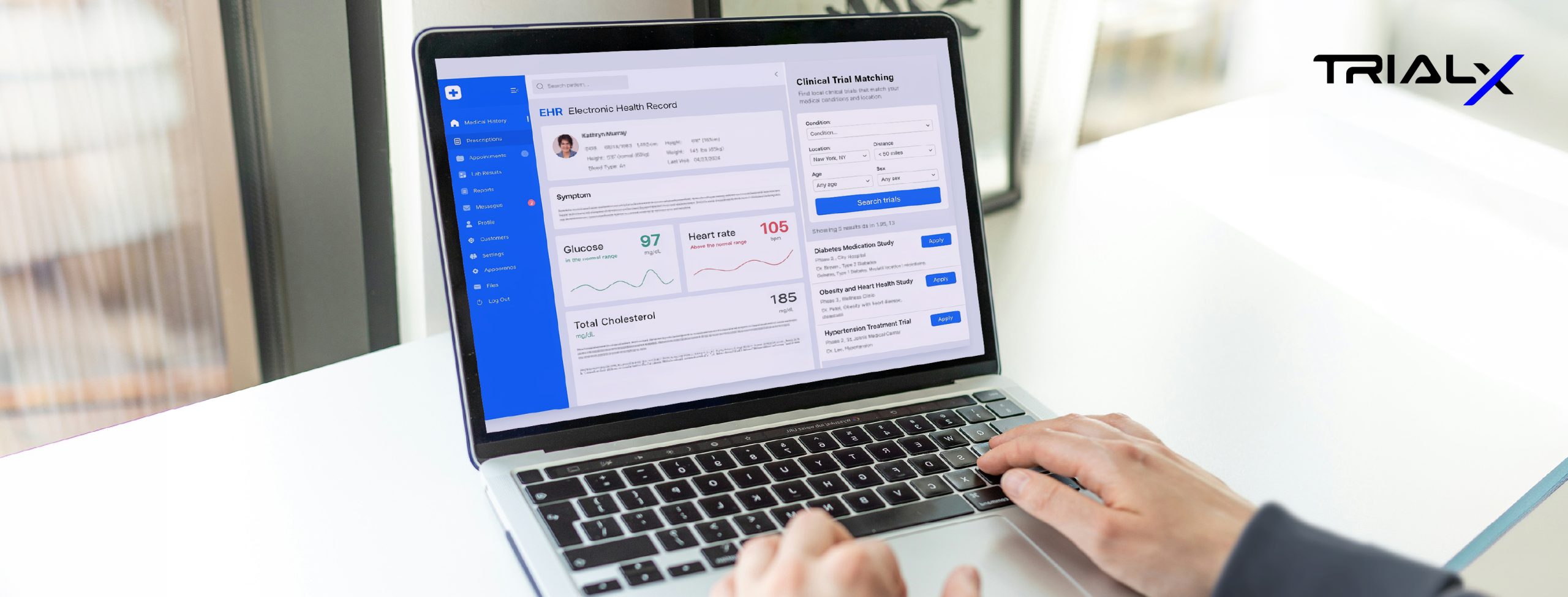
- Identifying potential participants earlier Matching tools can analyze patient records and highlight individuals whose clinical data may align with study criteria. This helps research teams become aware of relevant trials earlier in the care process. Within the TrialX platform, research teams can run structured prescreening workflows against incoming patient data, helping narrow the candidate pool before full screening begins.
- Reducing manual screening workload Instead of reviewing patient charts one by one, coordinators can focus on a smaller set of candidates whose records already show potential alignment with trial eligibility criteria. The platform also enables call center teams to request electronic medical records through secure links, allowing participants to authorize access to their records directly within the system—supporting eligibility assessment and recruitment coordination without added administrative friction.
For patients
- More relevant trial discovery Matching based on documented clinical information can help surface studies aligned with an individual’s health history. Through our smartEHR feature, patients can securely import their medical records and receive trial matches that reflect their specific clinical profile.
- Less reliance on manual searches Patients may be introduced to trials through the matching platform rather than manually locating studies themselves.
- Clearer starting points for eligibility discussions When matches are based on medical records, conversations about trial participation can begin with a clearer clinical context—reducing uncertainty on both sides of the conversation.
Together, these capabilities are designed to support coordinators, investigators, and patients in navigating trial eligibility more efficiently while helping research teams move toward their recruitment goals.