Rare Disease Awareness: Finding Tuberous Sclerosis Complex (TSC) Clinical Trials Through a Guided Search Experience

Rare Disease Day is observed every year on the last day of February. In the United States, a disease is considered rare (or orphan) when it affects fewer than 200,000 people. While each condition may impact a small population, nearly 7,000 known rare diseases together affect an estimated 300 million people worldwide.
Because patient populations are often small and only about 5% of rare diseases have an approved treatment—many individuals face long diagnostic journeys, limited care options, and less research investment compared to more common conditions. Despite these challenges, organizations such as TSC Alliance continue to play a critical role in advancing research, supporting families, and driving awareness for conditions that are often less visible.
Building on these efforts, we are proud to support the TSC Alliance through our AI-powered global clinical trial finder, helping connect patients and caregivers to relevant research opportunities. This collaboration supports the TSC Alliance’s mission to advance care and accelerate progress toward better treatments for Tuberous Sclerosis Complex (TSC)—a rare genetic condition affecting approximately 1 in 6,000 people at birth.
About Tuberous Sclerosis Complex (TSC)
TSC is caused by mutations in the TSC1 or TSC2 genes and can cause benign tumors to develop in vital organs, including the brain, kidneys, heart, lungs, and skin, with symptoms that vary widely from person to person. Some individuals experience mild symptoms, while others face more complex challenges, including epilepsy, kidney complications, and TSC-associated neuropsychiatric disorders (TAND).
Research into TSC has progressed over time, with FDA-approved therapies available for certain indications and ongoing studies exploring additional treatment approaches. However, for a condition like TSC — where the patient population is relatively small, and trials can be geographically dispersed — awareness and access at the patient level matter. Many families may not know which studies are recruiting, whether they or their loved one might qualify, or how to take the first step.
To help address this gap, the TSC Alliance Clinical Trial Finder, powered by TrialX, is designed to make clinical trial information easier to navigate for patients and caregivers, including those who may be new to clinical research. It offers two primary ways to explore studies.
1. Guided Search: A Step-by-Step Way to Find Relevant TSC Trials
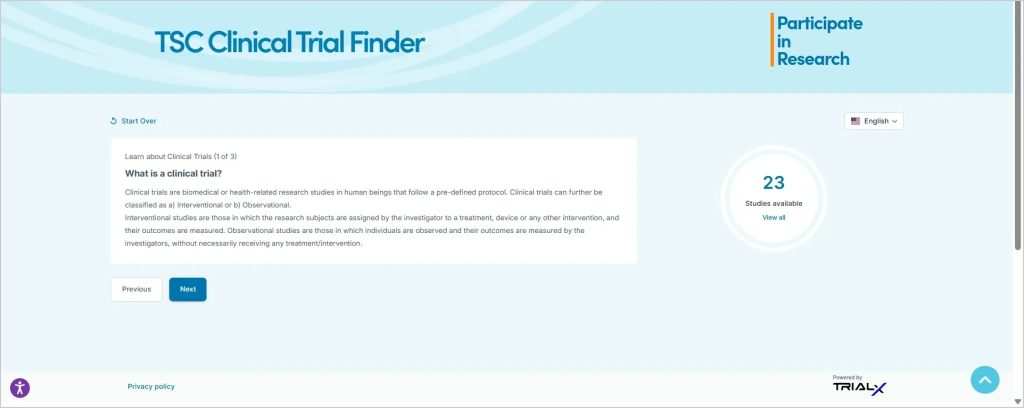
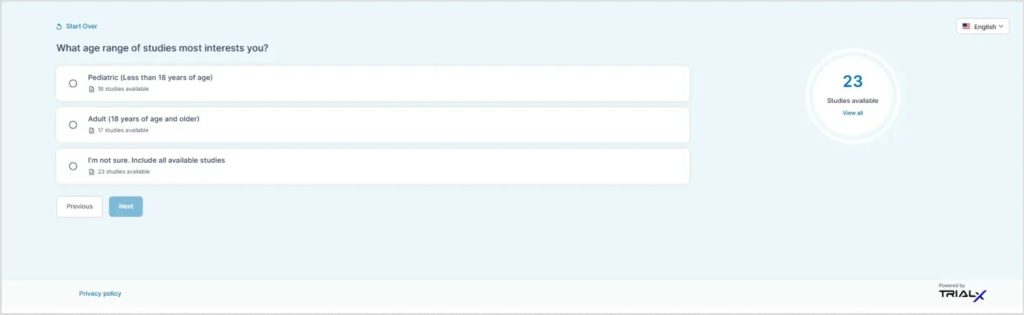
The guided search begins with brief, simple information about clinical research, including what participation may involve and what typically happens during a clinical trial. A clinical trial primer link directs users to a resource page that explains potential risks and benefits.
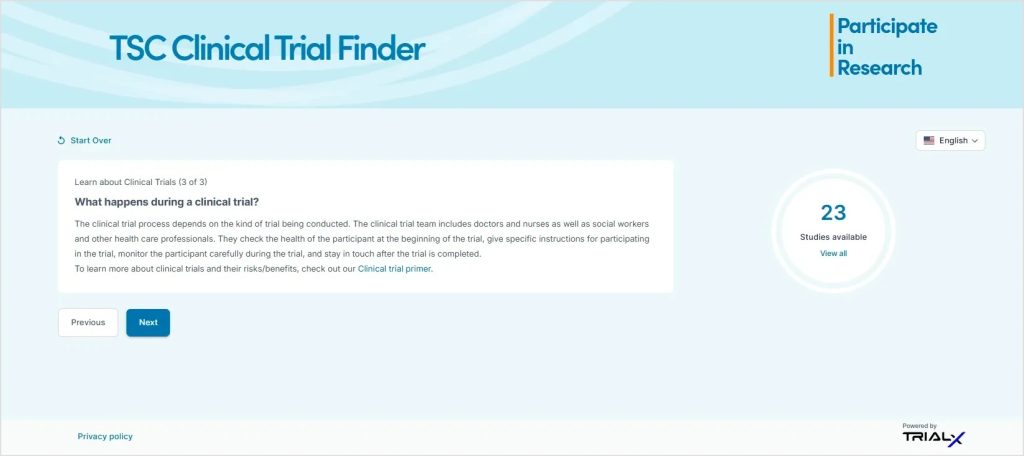
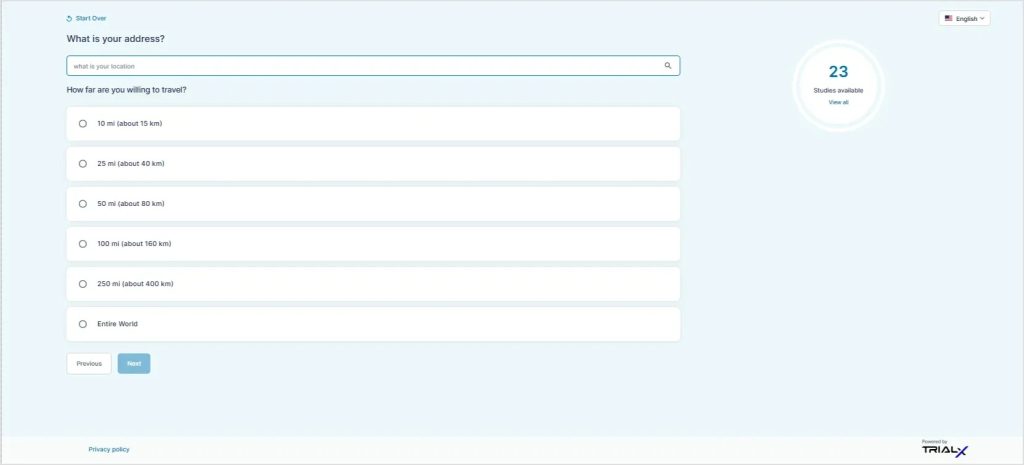
Users are then asked a few questions such as location, age, and willingness to travel. Based on these inputs, the finder presents clinical trials that may be relevant. This approach is intended to support individuals who may be less familiar with how clinical trials work.
2. Search On Your Own: Browse Available TSC Studies
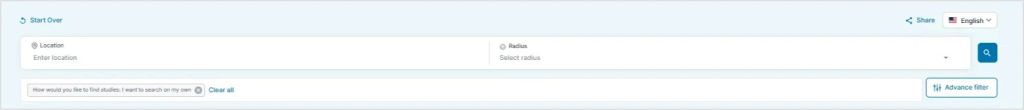
For those who prefer to explore independently, the trial finder also offers a Search on Your Own option.
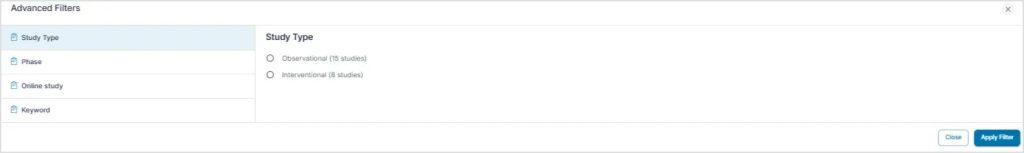
Users can view a list of available TSC studies and apply filters such as:
- Location
- Study type
- Trial phase
- Online or remote participation options
- Keywords
This option gives users more control to browse and compare studies at their own pace.
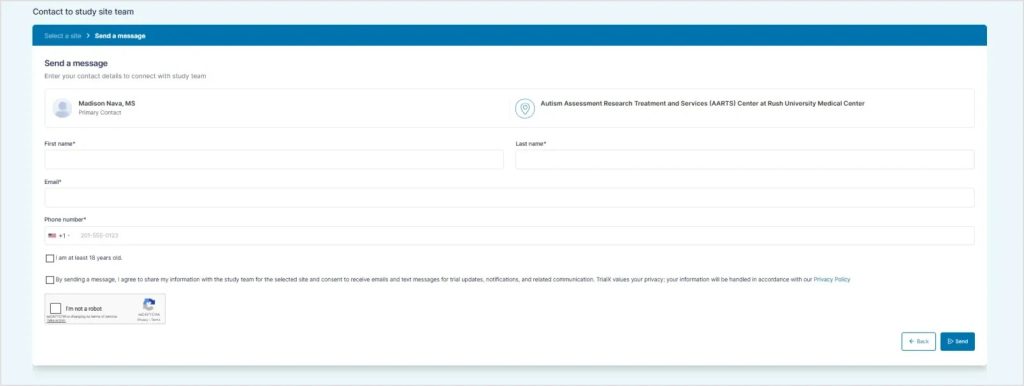
Clear contact options are included, allowing individuals to reach out to study teams directly if they want to learn more about a specific trial.
A Few Ongoing TSC Clinical Trials Currently Recruiting
Below are some important TSC-related studies that are currently recruiting participants:
Study of Skin Tumors in Tuberous Sclerosis
Sponsor- National Heart, Lung, and Blood Institute (NHLBI)
The study focuses on understanding why skin tumors develop in people with tuberous sclerosis. Adults aged 18 and older will have a skin exam with a dermatologist, and some may choose to have small skin samples taken using local anesthesia. These samples will help researchers study genetic changes and better understand how these tumors form and grow.
Location- Currently recruiting at the National Institutes of Health Clinical Center, United States.
Learn more about the study here.
Regulating Together (RT) Program for Youth with TSC
Sponsor- Cincinnati Children’s Hospital Medical Center (CCHMC)
This study is testing a program called Regulating Together (RT), which helps children with TSC learn how to better manage their emotions. Children will be placed into one of two programs: Regulating Together (RT) or a comparison program focused on school and organizational skills. The program runs for five weeks, with sessions held twice a week. Researchers will check in with families at several points to understand how well the program works over time.
Location– The trial is currently recruiting at Cincinnati Children’s Hospital Medical Center, United States.
Learn more about the study here.
ViRap: Preventive Treatment Study in Infants with TSC
Sponsor- Children’s Memorial Health Institute, Neurology and Epileptology
This study is testing two medicines—rapamycin and vigabatrin—to see which one works better and is safer when used early in infants with tuberous sclerosis complex (TSC). The goal is to help prevent or reduce symptoms such as seizures and tumor growth. Infants in the study will receive one of the two treatments and will be closely monitored over time. Researchers will track seizures, tumor growth, development, and any side effects to understand the long-term impact of early treatment.
Location- Currently recruiting at the Children’s Memorial Health Institute, Poland.
Learn more about the study here.
Why Clinical Trial Awareness Matters
“I started doing some online research and came across a doctor in Ohio who was doing a drug study on medicine that could help TSC patients. I was very fortunate to join the double-blinded drug study. I started the medicine in 2011, and in the first few months of being on the medicine, my tumors shrunk significantly. “
Misty’s experience shows how participation in clinical research can have a meaningful impact on people living with rare diseases like TSC. When patient populations are small, tools that simplify the process of exploring clinical trials—such as guided search—can help reduce uncertainty and make research information more approachable.
Rare Disease Day highlights the importance of awareness and participation in research. Increasing visibility and access to clinical trials helps move rare disease research forward and supports progress for the entire TSC community.
If you or someone in your family has TSC and would like to learn more about ongoing studies, the TSC Alliance Clinical Trial Finder provides information on current research opportunities.