Patient recruitment has always been an Achilles heel in the success of clinical trials. As Stuart Horowitz, President of Institutions and Institutional Services, WIRB-Copernicus Group cites “The greatest challenge for multicenter clinical trials is the timely recruitment of the required number of participants who complete studies and contribute evaluable endpoint data”
Patients are terrified to try anything new on their bodies. Investigators are already overworked. They have enough on their plates to be able to spend the required time with their patients to make them at ease with the complicated processes of a clinical trial or sometimes, even to let the patient know about a recruiting clinical trial. A bigger loophole is that, even though most patients trust their doctors and nurses most with clinical trial information, often times the care providers themselves are not aware of a recruiting clinical trial. These reasons largely account for as many as 45% of clinical trials finishing late and almost 80% failing to meet recruitment goal on time, adding to investigator burnout and harming the credibility of individual investigators and their institutions.
In the hope of encouraging recruitment in clinical trials, FDA recently updated its guidance to institutional review boards and clinical investigators clearly allowing reimbursements to patients in clinical trials for lodging and travel. This move is expected to ease things for the investigators, especially those who want to recruit people coming from other parts of the country to special treatment centers to participate in research.
While the effect of this updation is yet to be seen, what else is needed to help investigators attract more patients to their clinical trials?
Finding Solutions
Research shows that continued and clear communication is the key in attracting and retaining trial participants. So, the investigators, sites, and sponsors need a system in place to:
-
- Keep the patient well informed
- Present clinical trial information in the most easy-to-understand way possible
-
- Make it easy for patients to find clinical trials they are looking for
- Cut investigator’s management workload and spare them more time with the patients
Dr. Nina Bickell of Mount Sinai School of Medicine, a principal investigator involved in community engaged research says “The one constant in trial recruitment is it will always change, and you must adapt.”
In the past few years, digital methods are becoming more popular than traditional methods of advertisement in TV, radio and newspaper to spread the word and enroll patients owing to their ability to provide better presentation of information, more flexibility, and bigger outreach.
In an interview with Centerwatch, Sharib Khan, co- founder and chief executive officer of TrialX says “There isn’t one magic bullet to find patients for trials. We have created different tools and different ways to reach patients. You have to be available where patients are available.”
TrialX has designed an online platform iConnect 2.0 which helps academic medical centers, sponsors, and patient advocacy groups with patient-recruitment. The iConnect team has been providing support to major health organisations such as, BIDMC, NYU, New York Presbyterian, and Penn Medicine to meet their patient recruitment needs.
Since November 2017 iConnect 2.0 is also powering all clinical trial search at CenterWatch, which is most trusted by clinical trials professionals as well as patients, for any information related to clinical trials. As CenterWatch puts it, it has been the global destination for clinical trials information since its inception in 1994. It provides proprietary data and information analysis on clinical trials through their newsletters, books, databases, market research, benchmark reports and information services used by professionals involved in the management and conduct of clinical trials.
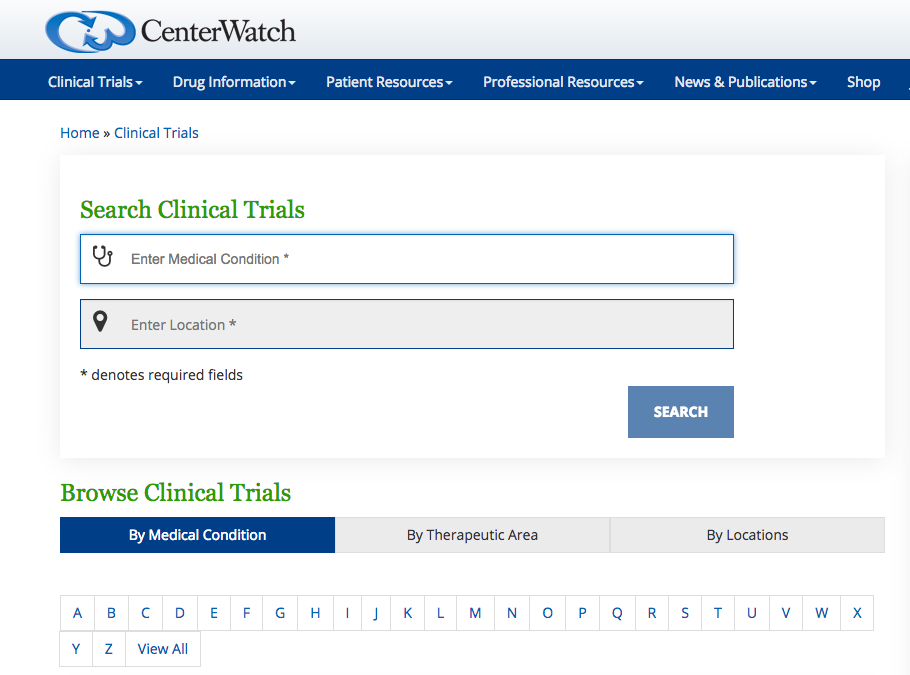
iConnect 2.0 has several features that the investigators can leverage to advertise their clinical trials better, save time and improve recruitment. Few of them are:
- Patient Friendly Clinical Trial Page to Aid Continued Communication: iConnect helps investigators make the content of a clinical trial page patient-friendly and editable without IT support. Once patients get the list of clinical trials, they can click on that trial link to check if the trial is still recruiting and if they are eligible for that trial, along with other specific details. Patients can contact the trial investigators at several sites, and write their concerns in a message, which is answered within 48 hours either via email or phone call, whatever the patient prefers.
- Clinical Study Specific Websites and Attractive Study Flyers as ToolKits: iConnect provides the investigator with options to help them build a clinical trial specific website and to make attractive flyers showcasing their trials. This feature helps researchers improve the layout and presentation of their trials so that its most comprehensible and inviting for patients. These websites and flyers would also serve as “physician toolkits” useful in building the knowledge base for referring doctors – hence aiding physician referrals.
- Online Outreach: iConnect also offers to increase online outreach of clinical trials helping patients and doctors be more aware of ongoing studies.
- Pre-screener to Save Investigator’s Time: This feature makes it easier for clinical trial investigators to identify the most appropriate patients for their trials. Researchers and research coordinators are usually approached by a lot of patients who are not eligible. This wastes a lot of their valuable time counselling and talking to these patients. The pre-screener helps researchers screen out the ineligible patients, cutting out their workload and time investment. At the same time it helps the patients too, in streamlining their search for a relevant clinical trial.
- 360 Degree Tracking with Real-time Recruitment Status Dashboard: TrialX iConnect also provides the investigators with a dashboard showing status of their trials, which can be edited with patient friendly descriptions and relevant and engaging images. Researchers can also get a trend of visitors over a period of time, such as, top trials viewed and top searches performed by visitors over a period of time by clicking on the Visitor Stats Tab. If researchers have advanced access to their institution’s iConnect, they can monitor and stay informed about the status of clinical trials in other departments in their institution. This helps researchers help their peers recruit patients for their clinical trials, making the process more efficient.
- Volunteer Registry as a Quick Reservoir for Upcoming Clinical Trials : If a patient approaches an investigator to participate in a clinical trial and he turns out to be non-eligible for that clinical trial, investigators have the option to put the details of that patient in a volunteer registry, so he can be notified and called when an appropriate trial becomes available.
With these and other advanced features iConnect aims to reduce investigator’s workload around clinical trial management, spare them more time with patients and facilitate patient recruitment for their clinical research.
The #SCOPE2018 opening plenary panel suggests that focus on patient recruitment and on making it easier for people to participate in clinical trials will continue as major trends in clinical research in 2018 and beyond. We look forward to more learnings towards increasing patient engagement and recruitment from the thought leaders sharing at SCOPE2018 in Florida this week.